KRAZATI Success in Treating Advanced KRASG12C-Mutated Lung Cancer

29 March 2024
Bristol Myers Squibb has announced successful outcomes from the KRYSTAL-12 Phase 3 trial. KRAZATI (adagrasib), targeting non-small cell lung cancer. It demonstrated significant benefits in progression-free survival and response rates compared to standard chemotherapy. Already FDA-approved, KRAZATI presents a promising option for previously treated patients. Ongoing studies aim to further confirm its efficacy and safety profile.
This confirmatory trial evaluated KRAZATI as a single-agent therapy in individuals with KRASG12C-mutated, locally advanced, or metastatic non-small cell lung cancer (NSCLC) who had previously undergone treatment. The study met its primary endpoint of progression-free survival (PFS) and a key secondary endpoint of overall response rate (ORR), offering new hope to patients.
Dr. Abderrahim Oukessou, VP and Global Program Lead for KRAZATI at BMS, expressed optimism, stating, “Today’s news is an important reinforcement of the power of a targeted therapy for patients with locally advanced or metastatic KRASG12C-mutated lung cancer. FDA approval of KRAZATI in the U.S. has allowed us to provide a new treatment option for these patients, and topline results of the KRYSTAL-12 confirmatory study will build greater trust in the medical and patient community.”
KRAZATI demonstrated a statistically significant and clinically meaningful benefit in terms of PFS and ORR, compared to standard-of-care chemotherapy. Notably, these patients had received at least one prior systemic therapy, positioning KRAZATI as a pivotal second-line or subsequent treatment option. Importantly, the safety profile of KRAZATI was consistent with previous findings, with no new safety concerns identified.
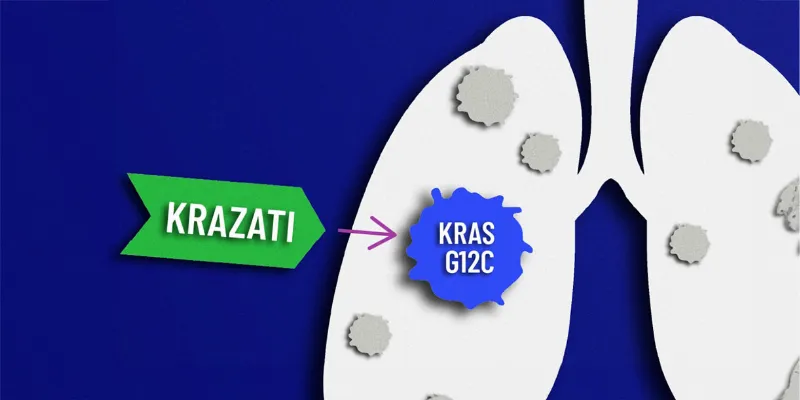
KRAZATI received accelerated approval from the FDA in December 2022 as a targeted treatment for patients with KRASG12C-mutated NSCLC who have previously received systemic therapy. Following this, MHRA and the European Commission granted conditional marketing authorization, further acknowledging KRAZATI's potential in addressing this challenging cancer mutation.
Beyond NSCLC, KRAZATI and its combinations are being explored in Phase 2 clinical trials for efficacy against various tumors, including advanced colorectal and pancreatic cancers. KRAZATI's ability to target and inhibit KRASG12C, a mutation present in a significant portion of NSCLC and other cancers, underscores the importance of precision medicine in oncology.









Comments
No Comments Yet!