TAR-210 Study Shows Promise in Bladder Cancer Treatment with 90% RF Survival Rate

5 May 2024
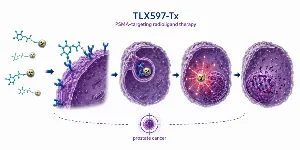
The TAR-210 study unveils groundbreaking findings in bladder cancer treatment, presenting a promising new approach for high-risk and intermediate-risk patients. Johnson & Johnson's innovative intravesical targeted releasing system, delivering erdafitinib locally, demonstrated impressive efficacy with a 90% recurrence-free survival rate and complete response rates.
Johnson & Johnson announced groundbreaking updated study results on TAR-210, indicating significant advancements in bladder cancer treatment. TAR-210, an intravesical targeted releasing system designed to offer sustained, local release of erdafitinib into the bladder, unveiled these findings in an Oral Presentation Session (Abstract # PD48-02) at the 2024 American Urological Association (AUA) Annual Meeting held in San Antonio, Texas.
These results offer hope for patients with limited treatment options, potentially reshaping the standard of care for non–muscle-invasive bladder cancer. Oral erdafitinib, known as erdafitinib (Balversa®), was approved by the U.S. FDA for patients with locally advanced or metastatic urothelial carcinoma (mUC) with susceptible FGFR3 genetic alterations.
Dr. Antoni Vilaseca, presenting author of the Phase 1 TAR-210 study from the Hospital Clínic de Barcelona, emphasized the significance of these findings, stating, "Advancement in the treatment landscape of high- or intermediate-risk non–muscle-invasive bladder cancer has remained stagnant for more than 50 years. Results presented today further underscore that TAR-210 for the localized treatment of bladder cancer may offer a promising alternative for patients with limited treatment options."
Bladder cancer remains a significant health concern, ranking as the sixth most prevalent cancer in the United States, with over 83,000 new cases diagnosed annually. Non–muscle-invasive bladder cancer (NMIBC) accounts for approximately 75-85% of these cases. Traditionally, treatment options for high-risk and intermediate-risk NMIBC have been limited, with standard therapies such as adjuvant intravesical immunotherapy with Bacillus Calmette-Guérin (BCG) or intravesical chemotherapy often proving insufficient.
However, the TAR-210 study offers a glimmer of hope for patients facing these challenges. The updated results highlight the potential of TAR-210 to revolutionize the treatment landscape for NMIBC, particularly in patients with fibroblast growth factor receptor (FGFR) alterations.
The study featured data from two key patient cohorts: Cohort 1 (C1), consisting of patients with recurrent, BCG-unresponsive high-risk NMIBC, and Cohort 3 (C3), comprising patients with recurrent, intermediate-risk NMIBC. Impressively, as of the data cutoff date of March 22, 2024, 64 patients had been treated with TAR-210 across the two cohorts. Among the 21 patients in C1 with HR-NMIBC, the 12-month recurrence-free (RF) survival rate was 90%. In C3, 31 patients were assessed for efficacy, revealing a complete response (CR) rate of 90%.
Importantly, TAR-210 exhibited a favorable safety profile, with the most common treatment emergent adverse events being Grade 1/2 lower urinary tract events. Moreover, there were no dose-limiting toxicities or treatment-related deaths reported during the study period.
Commenting on the significance of these results, Dr. Sabine Brookman-May, Vice President of Late Development Oncology at Johnson & Johnson Innovative Medicine, highlighted the potential of TAR-210 as a bladder-sparing and BCG-free treatment option. "FGFR genetic alterations are most common in NMIBC," she noted, "These results further support the potential of TAR-210 with quarterly administration as a bladder-sparing and BCG-free treatment option, underscoring our deep commitment to pioneering novel therapies for patients who face limited treatment avenues."
TAR-210 represents a promising investigational approach in the management of bladder cancer, offering renewed hope for patients with limited treatment options. As further research progresses, this innovative therapy holds the potential to reshape the standard of care for NMIBC, providing new avenues for improved outcomes and quality of life.







Comments
No Comments Yet!