AbCellera’s Breakthroughs in Antibody Discovery for Advanced Cancer Therapeutics


ONCOLife |
5 April 2024
In this exclusive interview for ONCOLife, we talk with Dr. Christopher Williamson and Tiffany Chiu from the biotech company AbCellera. They take us through AbCellera’s remarkable journey of innovation in antibody discovery and T-cell engagement therapies, shedding light on the company’s ambitious expansion of R&D endeavors towards pioneering cancer treatments.
Focusing on the challenges and potential of targeting peptide MHCs, GPCRs, and ion channels, Dr. Christopher Williamson and Tiffany Chiu provide a detailed overview of AbCellera’s approach to unlocking new classes of therapeutics. This conversation highlights not only its current achievements, but also sheds light on the exciting potential of AbCellera’s upcoming projects and the anticipated expansion of its drug pipeline.
Click the picture to view the PDF version: Pg 26-30.
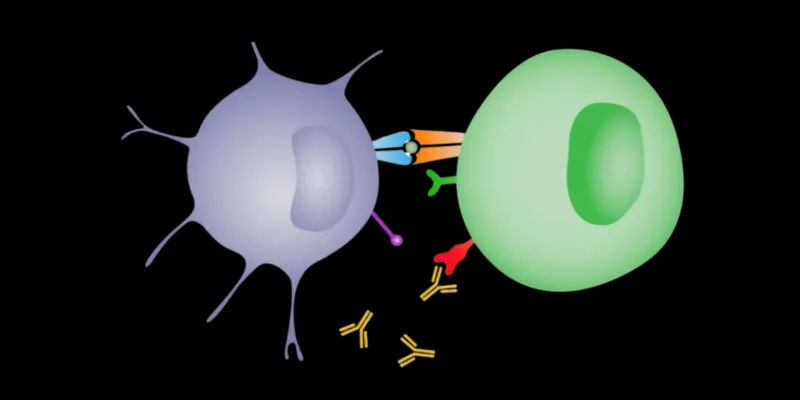
ONCOLife: AbCellera has developed a T-cell engager platform with a large panel of fully human CD3-binding antibodies. Could you elaborate on how this diversity in CD3-binding properties enhances the efficacy and safety of T-cell engagers compared to previous generations
Answer: T-cell engagers are emerging as promising new treatments in oncology, though their development has been hampered by complex design and engineering challenges. These therapies work by linking T-cells and tumor cells, enabling the immune system to destroy cancer cells. However, most traditional T-cell engagers rely on a non-proprietary CD3 binding arm discovered in the 1980s, which has been associated with dose-limiting toxicities, including high cytokine release.
To overcome these issues, our team has broadened the search beyond the traditional CD3 target. We’ve developed a variety of CD3 antibodies that bind to different CD3 sites with diverse affinities, offering a nuanced approach to engaging the immune system. This innovation aims to enhance the efficacy of T-cell engagers by increasing tumor cell destruction, while reducing adverse cytokine-related effects.
ONCOLife: AbCellera has developed a high-throughput process to streamline the creation TCEs for different tumor targets. Can you describe this process and how it prioritizes TCE function?
Answer: Our approach to generating TCEs involves combining our panel of CD3- binding arms with tumor-binding arms to develop bispecific antibodies. Identifying the most effective combinations requ-ires extensive testing. We create many different combinations of bispecifics, which are then subjected to thorough functional and biophysical assays to find those with optimal properties for development and functionality.
The challenge lies in determining which molecules will prove most effective. We prioritize identifying antibodies that strike a balance between effective tumor elimination and minimal cytokine release, alongside assessing their manufacturability and stability. This dual focus is essential for producing therapeutics that are both effective and viable for patient treatment.
ONCOLife: Aytokine Rele-ase Syndrome (CRS) is a significant concern with TCE therapies. How does your approach to selecting and optimizing CD3-binding antibodies minimize the risk of CRS, and what does this mean for the safety profile of your T-cell engagers?
Answer: Traditional T-cell engagers often overproduce cytokines, which, while killing tumor cells, also causes high levels of toxicity. There’s really no way to get completely away from uncoupling the therapeutic killing of the therapeutic with the cytokine release.
With our approach, we leverage our large CD3 panel to identify the most appropriate CD3 to pair with the most appropriate tumor-engaging arm in order to get the minimum cytokine release for the maximum cell killing. So, what we’re really trying to do is broaden the therapeutic index between where you see toxicity and where you see efficacy. And we believe that this may enable clinicians to dose at much higher levels and more effective concentrations.
ONCOLife: Your work on the MAGE-A4 x CD3 program targets a challenging peptide-MHC target. What makes MAGE-A4 particularly difficult to target, and how does your platform enable the development of effective TCEs against such targets?
Answer: Exploring the peptide MHC class of drug targets opens an exciting new space in T-cell engagers and therapeutics, allowing us to move beyond merely targeting surface proteins. This approach potentially unlocks the ability to target proteins expressed within tumor cells-areas previously inaccessible to treatments.
From a technical perspective, peptide-MHC proteins are a difficult class to get antibodies for. Our ability to find rare antibodies is something we believe gives us an advantage. Paired with our CD3 panel, this strategy aims to maximize cell killing while minimizing cytokines. This combination of tumor-targeting specificity with T-cell engaging efficacy seeks to broaden the therapeutic index, enhancing the potential for effective cancer treatments.
ONCOLife: With the advancement of your T-cell engager platform, what are the next tumor targets on your radar, and how are you prioritizing these targets based on clinical need and scientific opportunity?
Answer: We started working on TCEs in 2021 and have since launched five programs on a variety of tumor antigens, including peptide MHCs. We have shared our findings at both the American Association of Cancer Research (AACR) and the Society for Immunotherapy of Cancer (SITC) conferences. We’re presenting four more abstracts at AACR for this year’s annual meeting.
ONCOLife: Can this process be consistently used for other targets?
Answer: Over the last 12 years, we’ve been developing an engine, which includes the technology, expertise, and infrastructure to efficiently go from a drug target to clinical testing. We are using our antibody discovery and development engine across many targets and indications.
ONCOLife: AbCellera collaborates with various com-panies to advance novel science and technology. How do these partnerships enhance your ability to develop innovative immuno-oncology therapeutics, and what do you look for in a partner?
Answer: We have collaborated with major pharmaceutical companies, innovative biotechs, and venture capital groups who are building new companies. Currently, our focus is twofold. First, we are advancing our pipeline of programs by leveraging our robust capabilities to develop medicines independently—two programs are currently in IND-enabling studies. Second, we’re engaging in strategic partnerships with entities that offer novel biological or technological insights that complement our expertise, with a goal to bring new medicines to patients.
For instance, in late 2023, we announced a collaboration with Prelude to discover and develop precision antibody drug conjugates for oncology. Another example is our partnership with Abdera, which is advancing its precision radiotherapeutic for small cell lung cancer toward clinical trials. Both of these partnerships are examples of how we’re leveraging our combined capabilities and expertise to quickly discover and advance novel candidates into the clinic.
ONCOLife: Looking forward, what are AbCellera’s plans for expanding its research and development capabilities? Are there specific therapeutic areas or new technologies the company plans to explore beyond its current?
Answer: Our current focus is on enhancing downstream capabilities, which includes process development and manufacturing capabilities. Once complete, our engine will be able to cover the full spectrum, from discovery of a drug target to delivering potential new medicines to patients in Phase 1 clinical trials.
In terms of technology R&D, we’re focused on projects where we can open up new classes: T-cell engagers is one such area. We have also built technology to specifically tackle challenging targets called G protein-coupled receptors (GPCRs) and ion channels. These targets are linked to hundreds of diseases and have been notoriously difficult to address with antibodies. Of the two programs in our pipeline, one targets either a GPCR or an ion channel. Our strategy is to bring medicines to patients that are either first-in-class or best-in-class.
ONCOLife: What are your thoughts on the future of cell therapies over the next 5 to 10 years, particularly regarding their role in oncology?
Answer: Cell therapies, including CAR-T therapies, have been undergoing an interesting evolution over the past few years, validating the therapeutic approach of bringing a T cell to a tumor cell and then effectively getting an immune response: this approach overlaps with T-cell engaging bispecific antibodies. One of the challenges with cell therapies is the manufacturability, the cost, and the ability to scale these treatments. We believe these challenges can be surmounted with antibody therapeutics like T-cell engaging bispecifics.
General Overview of AbCellera
AbCellera (Nasdaq: ABCL), launched in 2012 by University of British Columbia academics, discovers and develops antibody medicines for indications across therapeutic areas including cancer, metabolic and endocrine conditions, and autoimmune disorders. AbCellera’s engine integrates technology, data science, infrastructure, and interdisciplinary teams to solve the most challenging antibody discovery problems. AbCellera is focused on advancing an internal pipeline of first-in-class and best-in-class programs and collaborating on innovative drug development programs with partners. For more information, please visit www.abcellera.com







Comments
No Comments Yet!