SUNRISE-II Begins First U.S. Trial of Ultrasound-Guided HIFU Ablation for Pancreatic Cancer
Key Takeaways
- Sonire’s first U.S. study of ultrasound-guided, anesthesia-free HIFU ablation therapy has been launched for patients with unresectable pancreatic cancer.
- The Stanford-led, open-label trial will enroll patients to assess safety, tolerability, and preliminary efficacy before first-line chemotherapy.
- This minimally invasive approach targets pancreatic tumors in a disease where the five-year survival rate remains about 13%.
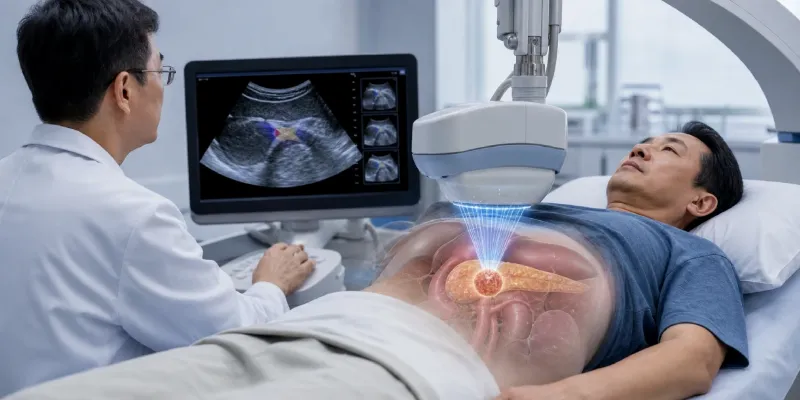
Sonire Therapeutics announced the initiation of SUNRISE-II, its first U.S. clinical trial evaluating Suizenji, an ultrasound-guided high-intensity focused ultrasound, HIFU, therapy system for patients with unresectable pancreatic ductal adenocarcinoma. The feasibility study, conducted at Stanford Cancer Institute, will assess safety, tolerability, technical performance, and preliminary clinical efficacy of this minimally invasive, anesthesia-free tumor ablation approach.
SUNRISE-II marks Sonire’s entry into U.S. clinical development and builds on the company’s prior experience in Japan. The study is open-label, non-randomized, and single-center, with 10 patients planned for enrollment. Eligible patients will have unresectable locally advanced or metastatic pancreatic ductal adenocarcinoma.
"Treatment options for pancreatic cancer remain extremely limited due to the complexity and location of the disease," said Tohru Satoh, President and CEO of Sonire Therapeutics. “Initiating SUNRISE-II in the United States is a defining milestone for our company. It enables us to begin building clinical evidence in a new market while advancing a treatment approach designed to be less invasive and more accessible for patients.”
The investigational device, Suizenji, uses focused ultrasound energy delivered from outside the body. Multiple ultrasound generators focus sound waves on a defined target within the pancreas. This concentrated energy heats the tumor area, causing thermal ablation of cancer cells. The system is guided by real-time ultrasound imaging, allowing physicians to monitor treatment delivery and target the tumor precisely.
A key feature of the platform is that it does not require general anesthesia. Prior experience with Suizenji in pancreatic cancer suggests that patients may perceive the heating effect as only a warm feeling in the abdomen during treatment. If confirmed in U.S. clinical testing, this could make the approach less burdensome for patients and potentially suitable for broader use in outpatient settings.
The first U.S. patient was treated in March 2026 at Stanford Medicine by Pejman Ghanouni, MD, PhD, principal investigator of SUNRISE-II. The trial will evaluate the device before initiation of standard first-line chemotherapy. All eligible participants will receive HIFU treatment on Day 1. Additional treatment sessions may be performed before chemotherapy if ablation is incomplete or judged insufficient by the principal investigator.
After post-HIFU assessments are completed, patients will begin first-line systemic therapy within two weeks of enrollment. Permitted chemotherapy regimens include FOLFIRINOX or modified FOLFIRINOX, gemcitabine plus nab-paclitaxel, or NALIRIFOX. All HIFU procedures and post-treatment assessments must be completed within this two-week window.
The primary objectives are to evaluate the safety and tolerability of Suizenji. Secondary objectives include assessment of overall device performance, technical success, and preliminary clinical efficacy. Participants will be followed at Month 1 and Month 3, with additional long-term follow-up until death or up to five years.
Although SUNRISE-II is small and exploratory, it addresses an important clinical question: whether noninvasive thermal ablation can be integrated safely into the early treatment pathway for patients with unresectable pancreatic cancer. For now, Suizenji remains investigational, and the study is not designed to prove survival benefit. However, the trial may generate the first U.S. feasibility evidence needed to support future regulatory submissions and larger clinical studies.
Pancreatic cancer remains among the most difficult malignancies to treat. Most patients are diagnosed at an advanced stage, and therapeutic options remain limited, especially for unresectable disease. According to the American Cancer Society, the five-year survival rate for pancreatic cancer is approximately 13%, underscoring the need for new local and systemic strategies that can improve outcomes without adding excessive treatment burden.



Comments
No Comments Yet!