FDA-Cleared Utepreva Device Aims to Improve Early Endometrial Cancer Detection
Key Takeaways
- The FDA 510(k)-cleared Utepreva Endometrial Sampler, a single-use device designed to support early diagnostic evaluation of endometrial cancer, may help improve diagnostic assessment.
- In preclinical verification testing, it captured more simulated tissue than a commercially available comparator and showed more uniform tissue disruption.
- The office-based procedure takes about 20 seconds, without dilation or sedation, supporting earlier diagnosis when survival may reach 95%.
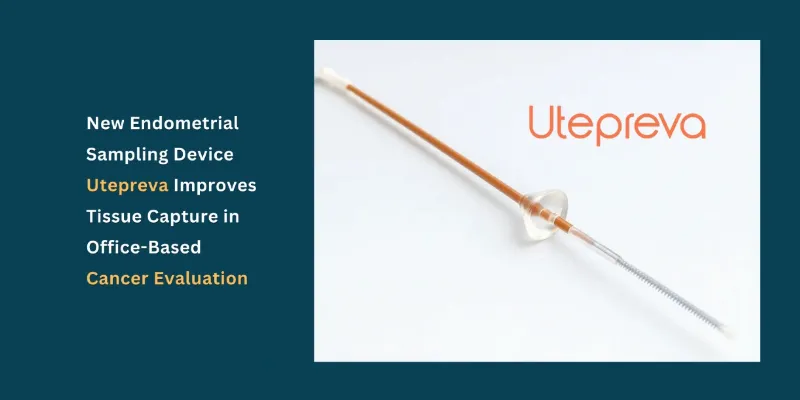
Utepreva has launched an FDA 510(k)-cleared, single-use endometrial sampling device designed to improve office-based evaluation of endometrial cancer by capturing both tissue and fluid in a single pass. The company says the Utepreva Endometrial Sampler was developed to address a persistent clinical problem: inadequate sampling with conventional devices, which can delay diagnosis and sometimes require repeat procedures.
The company positions the new sampler as a way to strengthen diagnostic evaluation at the earliest point of concern, particularly in symptomatic patients and women at elevated risk. The device is intended for use in patients with abnormal or postmenopausal bleeding, as well as those with risk factors such as older age, obesity, hormone exposure, or a family history of endometrial, colon, or ovarian cancer.
Unlike traditional endometrial samplers that rely mainly on disruption or suction alone, the Utepreva device combines three mechanisms: gentle tissue disruption, controlled suction, and absorption through a sponge tip. This design is intended to improve tissue yield and sample the uterine cavity more comprehensively. The collected material may support cytologic, histopathologic, and molecular analyses, potentially allowing both routine cellular assessment and more advanced biomarker evaluation from one specimen.
The company cited preclinical and design verification testing conducted by Medical Murray, in which the Utepreva device was compared with a commercially available endometrial sampler using a standardized simulated tissue model. Under controlled conditions, Utepreva reportedly captured a greater volume of simulated tissue and produced more uniform tissue disruption across the sampling surface, with statistically significant performance versus the comparator device.
"Our mission is to save lives by expanding access to minimally invasive uterine evaluation and aiding clinicians in the detection of pre-cancer, when treatment is less invasive, and survival outcomes are significantly improved," said Dr. Jeanetta Stega, CEO of Utepreva.
The company also points to a broader ambition. While the device is currently recommended for symptomatic and high-risk patients, it may eventually have a role in routine gynecologic care if further evidence supports broader use.
"As physicians, we've seen firsthand how improved screening tools revolutionize patient outcomes," said Dr. Jeanetta Stega. “By dramatically improving tissue capture in a simple office procedure, Utepreva aims to make a similar impact on pre-cancer detection to that of Pap smears and mammography. As clinical evidence continues to build, enhanced sampling technology could ultimately lay the groundwork for broader, earlier evaluation strategies in the future.”
Uterine cancer is the most common gynecologic malignancy in the United States, and most cases are endometrial carcinoma. According to figures cited in the announcement, an estimated 68,270 new uterine cancer cases will be diagnosed this year. Outcomes remain closely tied to stage at diagnosis. Survival can reach as high as 95% when disease is detected early, but drops to below 20% in advanced disease.





Comments
No Comments Yet!