Rznomics' RNA-Based Therapy Receives FDA Orphan Drug Status for Liver Cancer

FDA & EMA |
2 February 2024
Rznomics has received FDA Orphan Drug Designation for RZ-001, a novel RNA-based therapy for Hepatocellular carcinoma. RZ-001 employs advanced RNA editing to target and suppress cancer growth, showing promise in preclinical studies.
The South Korean biopharmaceutical company Rznomics Inc. has announced that its novel RNA-based therapy, RZ-001, designed for the treatment of Hepatocellular Carcinoma (HCC), has been granted Orphan Drug Designation (ODD) by the US Food and Drug Administration (FDA). Rznomics is also collaborating with Roche to study RZ-001's efficacy in combination with atezolizumab.
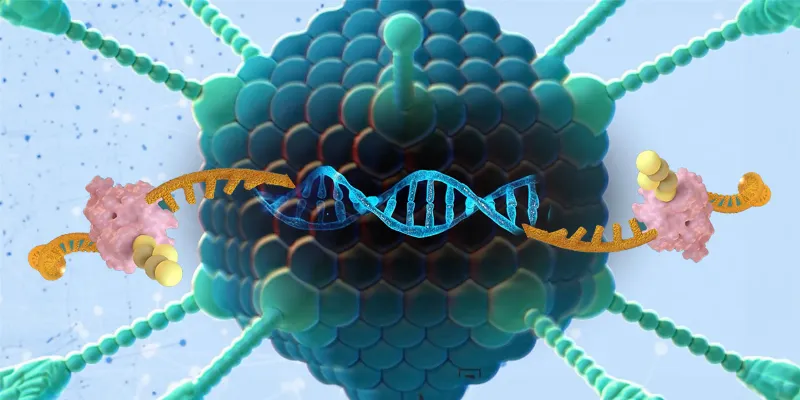
RZ-001 is a trans-splicing ribozyme derived from the self-splicing Tetrahymena group I intron, offering unique features that differentiate it from other nucleic acid-based editing approaches. The technology involves a single RNA molecule capable of both suppressing target RNA expression and simultaneously expressing therapeutic RNA, potentially eliminating the need for additional proteins or cofactors.
Safety is enhanced by selectively inducing therapeutic RNA expression only in cells where the target gene is expressed and by regulating the expression proportionally to endogenous cellular target RNA levels. Importantly, editing occurs at the RNA level, avoiding concerns about genomic toxicity and permanent genome changes.
Seong-Wook Lee, CEO and founder of Rznomics, expressed enthusiasm about the FDA Orphan Drug Designation, stating, "This FDA Orphan Drug Designation further underlines the potential of our pipeline to expeditiously address the current unmet medical needs of patients with Hepatocellular Carcinoma."
RZ-001 specifically exhibits effective anti-HCC activity by selectively suppressing hTERT expression in cancer cells, inducing a cytotoxic effect by trans-ligating an HSVtk-encoding sequence into the reprogrammed hTERT mRNA. This innovative approach has shown promising results in preclinical animal models, inducing immune cell infiltrations into HCC tumors.
Rznomics has obtained Phase I/IIa Investigational New Drug (IND) approvals for RZ-001 in Hepatocellular Carcinoma from both the FDA and the South Korean Ministry of Food and Drug Safety (MFDS). The company plans to conduct a dose escalation/expansion study to investigate the safety, tolerability, and efficacy of RZ-001 in HCC patients with no extrahepatic metastasis. Additionally, Rznomics has received Phase I/IIa IND approvals for RZ-001 in Glioblastoma Multiforme (GBM) in both Korea and the U.S., with a Fast Track designation.
In further development, Rznomics has entered into an HCC clinical collaboration agreement with F. Hoffmann-La Roche Ltd (Roche) to study RZ-001 in combination with Roche's atezolizumab. This collaboration highlights Rznomics' commitment to advancing innovative treatments for Hepatocellular Carcinoma.
FDA Orphan Drug Designation
Orphan Drug Designation is a crucial milestone for RZ-001, potentially paving the way for seven years of market exclusivity upon approval, along with significant regulatory and financial incentives such as exemption from certain fees and tax credits. It also facilitates access to specialized advice from the FDA's Office of Orphan Products Development, a boon for treatments targeting rare diseases.





Comments
No Comments Yet!