Light-Activated Copper Nanoparticles Trigger Cuproptosis in Drug-Resistant Cancer Cells
27 March 2026
A light-activated copper nanoparticle platform induced cuproptosis and killed drug-resistant human breast cancer cells at nanomolar concentrations, offering a potential alternative to apoptosis-based chemotherapy. The approach was reported to be about 100 times more effective than existing platinum drugs, while remaining inactive in the dark, a design that may reduce off-target toxicity and improve treatment precision.
Cancer therapy has long depended on a familiar playbook: deliver a cytotoxic drug systemically, hope enough of it reaches the tumor, and accept that healthy tissue may be caught in the crossfire. A new study from researchers at Ruhr University Bochum suggests there may be another route, one that relies on a recently identified form of regulated cell death and activates only when illuminated.
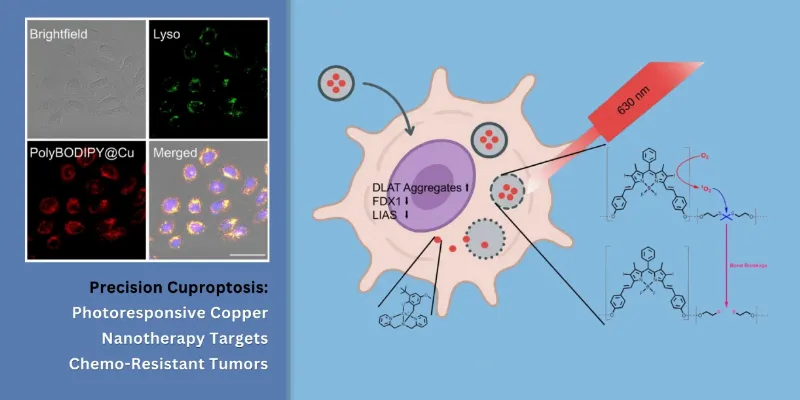
The approach centers on cuproptosis, a copper-dependent mechanism first described in 2022. Unlike apoptosis, which underpins many conventional chemotherapies, cuproptosis appears to attack a tumor’s metabolic core. In the new work, published in Advanced Functional Materials, investigators designed a photo-responsive nanoparticle system that stays inactive in the dark but releases a copper-based cytotoxic payload after exposure to red light. In resistant human breast cancer cells, the platform produced potent anticancer activity in the nanomolar range.
That matters because resistance remains one of chemotherapy’s most stubborn problems. Many anticancer drugs ultimately fail not because they cannot kill cancer cells, but because tumors adapt to the death pathways those agents are built to exploit. The Bochum team took aim at that limitation by triggering a non-apoptotic route.
Cancer cells often handle copper differently from healthy cells, in part because of their altered metabolic demands. As Prof. Dr. Johannes Karges, from the Department of Biophysics, Faculty of Medicine, Ruhr University Bochum, and one of the authors of the study, explains, “What makes this type of cell death so unique is its specificity in targeting the cell’s energy production. Cancer cells often have an altered, particularly intense metabolism and take in more copper than healthy tissue does.”
The engineered system combines several layers of control. At its core is a tripodal copper complex, the active cytotoxic agent. This is packaged inside amphiphilic polymeric nanoparticles that self-assemble into stable spherical structures, with a hydrophobic interior carrying the drug and a PEG-functionalized exterior that improves colloidal stability and biocompatibility. Embedded in the polymer backbone is a BODIPY photosensitizer, along with a reactive oxygen species-sensitive linker.
In practical terms, the nanoparticles remain intact under physiological conditions without light exposure. Once irradiated with red light, the BODIPY unit generates singlet oxygen, which oxidizes the linker and causes the nanoparticle to disassemble. The copper complex is then released locally, rather than systemically.
Prof. Dr. Karges describes the mechanism succinctly: “The release principle is based on a photo-responsive bond within the basic polymer framework. Light radiation selectively severs this specific bond, whereupon the nanoparticles dissolve and the copper complex is released locally.”
This design gives the platform both spatial and temporal precision. The particles are meant to accumulate preferentially in tumor tissue, while the polymer shell helps prevent premature release. Only after targeted light exposure does the therapeutic switch turn on. In principle, that could reduce off-target toxicity, a central weakness of standard chemotherapy.
Mechanistically, the released copper complex induces cuproptosis by disrupting regulators of mitochondrial metabolism, including lipoic acid synthetase and ferredoxin 1, while promoting oligomerization and aggregation of dihydrolipoamide S-acetyltransferase. The end result is severe proteotoxic and metabolic stress, driving cell death through a pathway distinct from those commonly engaged by platinum-based drugs.
The authors report that the platform was roughly 100 times more effective than clinically used platinum derivatives in their experimental setting. Just as important, activity was maintained in multidrug-resistant cancer cell derivatives, suggesting that the strategy may bypass resistance mechanisms that blunt the effect of conventional agents.
As Prof. Karges notes, “We were also able to show that this method is effective in treatment-resistant cancer cells, where conventional chemotherapy treatments hit their limits.”
Still, the findings remain firmly preclinical. The work was conducted in resistant cancer cells in the laboratory.
Prof. Karges is careful not to overstate the translational timeline: “So far, we have shown this on resistant cancer cells in the lab, not in a human body. A lot still has to be done before an actual treatment can be performed.”
Even so, the study points to a broader shift in oncology drug design. Instead of asking only how to kill cancer cells more powerfully, researchers are increasingly asking how to do it more selectively, more controllably, and through mechanisms tumors are less prepared to evade.









Comments
No Comments Yet!