New Protein Modification Linked to IDH-Mutant Cancers Opens Therapeutic Possibilities
26 March 2026
A Nature Chemistry study identifies a previously unknown protein modification, O-2-hydroxyglutarylation, driven by the oncometabolite D2HG in IDH-mutant cancers. Researchers also found distinct chiral protein modifications by D2HG and L2HG, with effects on kinases MRCKA and SLK and reduced substrate phosphorylation. The findings reveal a new link between altered metabolism and cancer signaling, with potential therapeutic implications.
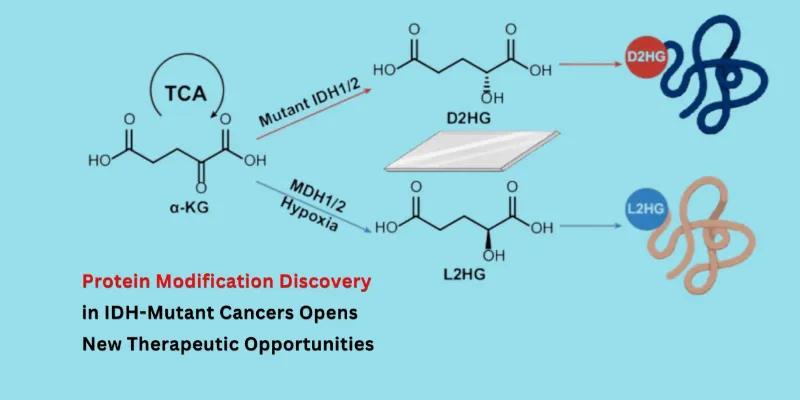
Cancer researchers have spent years studying mutant IDH enzymes because of their central role in tumor metabolism. These mutations are especially common in gliomas, acute myeloid leukemia, and some rare liver cancers, where they rewire a core biochemical pathway and cause cells to accumulate D-2-hydroxyglutarate, or D2HG, instead of the normal metabolite alpha-ketoglutarate. Now, a new study suggests that D2HG may do more than build up inside cancer cells. It may also leave a direct chemical mark on proteins, altering how those cells function.
In a paper published in Nature Chemistry, a team led by Purdue University’s W. Andy Tao reported the discovery of a previously unknown protein modification called O-2-hydroxyglutarylation. The finding adds a new layer to the biology of IDH-mutant cancers and points to a possible therapeutic opportunity beyond simply blocking the mutant enzyme itself.
“Mutation is considered the driving mechanism leading to cancer. Many mutations are hidden and harmless, but the mutation of enzymes like kinases can lead to the uncontrolled growth of cancer cells,” said Tao, a professor of biochemistry in Purdue’s College of Agriculture.
Under normal conditions, IDH enzymes help convert isocitrate into alpha-ketoglutarate, a key step in cellular energy metabolism. But when IDH1 or IDH2 is mutated, that reaction is derailed, and D2HG accumulates. This oncometabolite has already been linked to impaired cell differentiation and tumor progression. The new study shows that it can also chemically modify proteins in a way that appears to influence kinase signaling.
Using chemical proteomics, the researchers found that D2HG-driven protein modifications were increased in IDH-mutant cells and after D2HG treatment. They also found that the related metabolite L-2-hydroxyglutarate, or L2HG, produced its own distinct pattern of protein modification. That difference matters because D2HG and L2HG are chiral molecules, mirror images of one another that can behave differently in living systems.
“People haven’t thought about it. And, certainly, few have reported chiral-dependent modification of metabolites,” Tao said.
The distinction may be particularly relevant in cancer. D2HG accumulates in IDH-mutant tumors, while L2HG tends to increase under hypoxic conditions, another hallmark of aggressive solid tumors. The study found that the two metabolites target different kinases: MRCKA was modified by D2HG, while SLK was modified by L2HG. Synthetic peptide standards confirmed both events. Phosphoproteomic analysis then showed reduced phosphorylation of downstream substrates, suggesting that these metabolite-driven protein marks can interfere with kinase activity.
That observation is important because phosphorylation is one of the cell’s most important signaling mechanisms, especially in cancer. “Phosphorylation is important because many phosphorylated proteins are messenger molecules related to cancer,” Tao said. “People usually do not consider this IDH mutation related to phosphorylation, but by identifying this modification, we see crosstalk with phosphorylation.”
In laboratory analyses of normal cells, IDH1-mutant cells, and IDH1-mutant cells treated with an anticancer drug, Tao’s team identified dozens of D2HG-modified proteins. Many were linked to tumor progression. As the authors wrote, “Many of these proteins are involved in tumor progression, suggesting that D2HG modification may play a regulatory role in cancer development,”
That is what makes the work more than a biochemical curiosity. Post-translational modifications, chemical edits made to proteins after they are produced, are already central to modern drug development. If D2HG- and L2HG-driven modifications help sustain malignant signaling, they could become targets in their own right.
“IDH mutation is quite common in multiple cancer cells, especially for glioma, acute myeloid leukemia and rare liver cancer,” Tao said. “By identifying this new modification, there’s potential to open a new path for therapeutics,” Tao said.
Mutant IDH may not simply poison metabolism, it may reprogram the signaling machinery of the cancer cell itself. That expands the field’s understanding of how oncometabolites drive disease, and suggests that the next generation of therapies may need to target not only the mutation, but also the protein chemistry it leaves behind.









Comments
No Comments Yet!