Abenacianine Enters Phase 3 to Enhance Real-Time Tumor Detection in Lung Surgery

19 March 2026
Vergent Bioscience has launched the Phase 3 VISUALIZE 2 trial evaluating abenacianine, a tumor-activated imaging agent designed to enhance intraoperative tumor detection in lung cancer surgery. The study will enroll 132 patients across 10 sites. Earlier trials showed improved surgical decision-making in up to 45% of cases, highlighting its potential to improve resection precision.
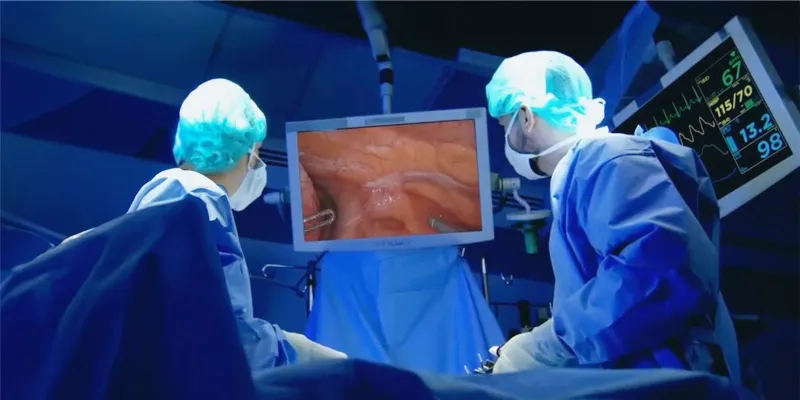
The multi-center study will assess whether the agent can enhance detection of malignant tissue, particularly in minimally invasive and robotic-assisted procedures where tactile feedback is limited.
Surgical resection remains a cornerstone of treatment for lung cancer and other solid tumors. While minimally invasive and robotic approaches have improved patient outcomes, including reduced trauma and faster recovery, they also restrict surgeons’ ability to directly see and palpate tissue. This limitation can complicate accurate tumor localization and complete resection.
Abenacianine is engineered to address this challenge by illuminating cancerous tissue in real time during surgery. The agent is tumor-activated and uses near-infrared imaging to reveal lesions that may not be visible under standard white light. In earlier Phase 2 and 2b studies, the technology contributed to improved intraoperative decision-making in 43 percent and 45 percent of procedures, respectively.
“We are excited to advance abenacianine into this next stage of clinical development, working with leading thoracic surgeons, academic institutions, and community cancer centers,” said John Santini, Ph.D., president and chief executive officer at Vergent Bioscience. “By revealing malignant tissue undetectable by white light, abenacianine brings molecular intelligence into the operating room, helping surgeons make more confident decisions when precision matters most. We believe abenacianine has the potential to become critical operating room technology as surgery continues to evolve and become more minimally invasive, robotic, and precise.”
The VISUALIZE 2 trial is a randomized, open-label study expected to enroll 132 patients across 10 sites in the United States and Australia in 2026. Participants will receive a single intravenous infusion of abenacianine 12 to 96 hours prior to surgery. During the procedure, surgeons will first rely on standard techniques to identify lesions, followed by near-infrared imaging to detect additional tumor tissue. All findings will be confirmed through histopathological analysis.
The primary endpoint focuses on the proportion of patients with at least one Clinically Significant Event, defined as the identification of previously undetected lesions or additional cancer not found using conventional methods.
“Once surgery begins, even the most carefully planned procedures can sometimes be limited by what is visible under white light,” said Sunil Singhal, M.D., principal investigator of the study. “By turning light into molecular insight, abenacianine aims to provide surgeons with actionable, real-time information, and we look forward to confirming if it supports more precise, tissue-sparing decisions.”
Mechanistically, abenacianine targets cathepsins, proteases commonly overexpressed in solid tumors, and incorporates indocyanine green, a near-infrared dye compatible with widely used surgical imaging systems. This design enables integration into existing workflows without requiring additional infrastructure, potentially facilitating adoption if clinical benefit is confirmed.









Comments
No Comments Yet!